Optimization of hematite increases photoelectrolyzer efficiency for green hydrogen production
CINE researchers took an important step forward in the development of an efficient photoelectrolyzer for the generation of green hydrogen.
Photoelectrolyzers are equipment that split the water molecule (H2O) using sunlight as an energy source. Their main components are the photoanode and the photocathode, which absorb light and transform it into charges. When they reach the surface of the material, these charges promote oxidation and reduction reactions of water, which generate, respectively, oxygen (O2) and hydrogen (H2).
These devices offer a simple, low-cost, sustainable and clean way to produce hydrogen. However, the technology is still not competitive, and one of the main challenges to make it commercially viable is to find a material that efficiently acts as a photoanode, converting light energy into electrons capable of oxidizing the water molecule.
In this new work, the authors designed a strategy to optimize hematite (Fe2O3), a very abundant iron oxide which is maybe the most used material in photoanodes. The advance led to a 50% increase in photoelectrolyser efficiency.
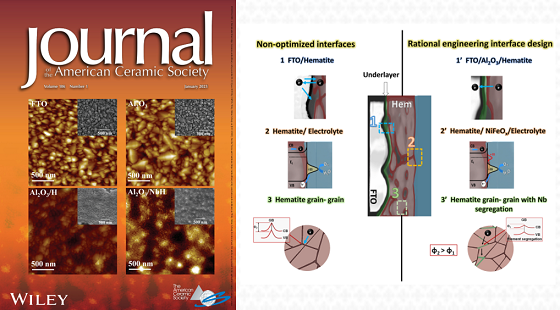
To arrive at this result, the researchers focused their attention on the interfaces where the greatest loss of electrons occurred: between hematite and its substrate, among hematite grains, and between hematite and water. The authors modified the hematite with elements that are also abundant, such as niobium and aluminum, through a simple manufacturing process, capable of manipulating all these interfaces.
The optimized photoanode generated an electrical current 6.7 times greater than the conventional hematite photoanode. “We believe that this research was a great step towards the development of the first national prototypes for the production of green hydrogen via photoelectrolysis”, says Flavio Leandro de Souza, professor at UFABC and coordinator of the green H2 program at LNNano/CNPEM, who led the work.
“The big challenge was to control the interfaces to guarantee that the energy of the light absorbed by the material is used to promote the chemical reactions that culminate in the generation of H2”, says Souza, who is a researcher in the Dense Energy Carriers program at CINE. “These interfaces prevented or made it difficult for the charges to reach the surface of the material that is in contact with the water”, he adds.
According to him, the modified iron photoanodes still have limitations that make necessary to apply an external voltage in the system to complement the photogenerated current. However, in order to maintain the “green” character of hydrogen, it is possible to use photovoltaic or wind energy, or even to integrate solar concentrators into the photoelectrolyzer.
The work was featured on the cover of a special volume, dedicated to materials for the energy field, of the traditional Journal of the American Ceramic Society.
Conducted at LNNano/CNPEM, with the collaboration of students and researchers from UFABC and the International Research Center for Renewable Energy (China), the study received funding from FAPESP, Shell, CAPES, CNPq, NSFC (China), as well as strategic support of ANP.
Paper reference: On electron loss lowering at hematite photoelectrode interfaces. Karen C. Bedin, Ingrid Rodríguez-Gutiérrez, Lizandra R. P. Peregrino, Lionel Vayssieres, Flavio L. Souza. J Am Ceram Soc. 2023; 106:79–92. DOI: 10.1111/jace.18460
Paper authors who are members of CINE: Karen C. Bedin (postdoc in the Dense Energy Carriers program), Flavio L. Souza (principal researcher in the Dense Energy Carriers program).
Contact

Flavio Leandro de Souza
UFABC - LNNano

