New material improves the performance of sodium-ion batteries and supercapacitors
CINE researchers and collaborators have developed a material capable of improving the performance of batteries and supercapacitors based on sodium ions – a promising alternative to lithium-ion technology, which is currently the most used.
In these technologies, lithium or sodium ions are the protagonists, as they are responsible for transporting and storing electrons during the charge (energy storage) and discharge (energy delivery) processes. Given that the ions penetrate the material structure of the electrodes in charge and discharge, it is important that they are small. At this point, lithium is the champion, being the smallest solid-state ion in the periodic table. Sodium is also among the smallest ions, but its main advantage is the abundance and good distribution of this element on the planet, both in the earth’s crust and, mainly, in the sea. In the case of lithium, reserves are less abundant and are concentrated in a few countries.
“In our work we have developed devices that work with sodium ions – a technology whose economic viability is much more interesting than that of lithium ions and which could be manufactured in many countries, including Brazil”, says Professor Hudson Zanin (UNICAMP), researcher at CINE’s Advanced Energy Storage program.
This technology, however, is not yet ready for commercialization, and one of the main reasons is the fact that the speed of insertion of ions into the electrodes and their exit from them is slower due to the larger size of the ions. Therefore, the development of electrodes that increase this speed is one of the main challenges to make the sodium-based technology viable.
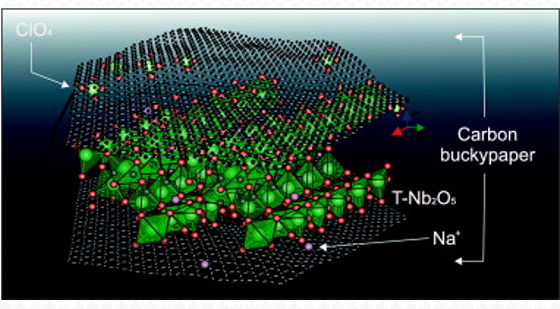
In a recently published article in the Journal of Energy Storage, Zanin and the other authors present a new material, formed by carbon nanotubes with niobium pentoxide nanoparticles ( NbO5), for the electrodes of that kind of device. The team studied the performance of the material as the electrode of a sodium-ion battery and of a sodium-ion supercapacitor.
The results of experiments and computer simulations showed that the electrode worked very well in the main charge storage mechanisms: intercalation and adsorption. The authors observed that the sodium ions intercalated into the niobium pentoxide at an ultra-fast rate and that they were well adsorbed by electrostatics in the large surface area of the carbon nanotubes. As a result, the devices’ capacitance (their ability to collect and store electrical charges) increased significantly over the conventional nanotube electrode without nanoparticles.
The study was carried out as part of Carla Real’s PhD in Electrical Engineering, which is underway at UNICAMP with the advise of Professor Zanin, and had the collaboration of researchers from Kansas State University (USA) in the production and characterization of the sodium battery. Researchers from the Federal University of Mato Grosso (UFMT) and the Federal University of Vales do Jequitinhonha e Mucuri (UFVJM) also participated in the work, helping to understand the fundamental electrochemical phenomena that occurred in the devices.
FAPESP, Shell, CNPq and Capes financed the research, and the ANP provided strategic support.
Scientific article reference: Freestanding niobium pentoxide-decorated multiwalled carbon nanotube electrode: Charge storage mechanism in sodium-ion pseudocapacitor and battery. Carla G. Real, Ericson H. N. S. Thaines, Leandro A. Pocrifka, Renato G. Freitas, Gurpreet Singh, Hudson Zanin. Journal of Energy Storage. Volume 52, Part B, 15 August 2022, 104793. https://doi.org/10.1016/j.est.2022.104793.
Authors of the article who are members of CINE: Carla Real (PhD student at UNICAMP), Renato Freitas (professor at UFMT) and Hudson Zanin (professor at UNICAMP).
Contact

Hudson Zanin
UNICAMP - Brasil

