CINE’s computational study contributes to the development of more selective catalysts for the transformation of CO2 into raw materials for the industry.
Known by the acronym CCUS, the concept of “Carbon Capture, Utilization and Storage” covers the processes and technologies which aim at reducing the concentration of carbon dioxide (CO2) in the atmosphere through the safe storage of that greenhouse gas or its transformation into other molecules which can be used as raw material for the fabrication of fuels and other products.
In a recently published paper, CINE researchers present a contribution to this set of technologies; more precisely, to the electrochemical reduction of CO2, a process used to transform carbon dioxide into compounds of industrial interest such as carbon monoxide (CO) or formic acid (HCOOH).
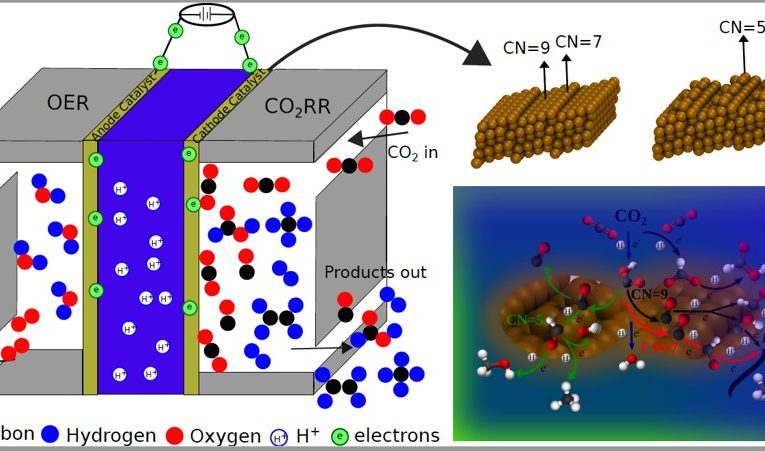
“For this process to occur efficiently and be financially viable, one of the challenges which need to be overcome is the development of catalysts for CO2 reduction which are more active, selective (capable of forming as a majority a product of interest) and durable”, says post-doc Lucas Garcia Verga, the first author of the article. In this context, catalysts of cooper (Cu), which is a low cost and low environmental impact metal, gain prominence for generating, efficiently, molecules of industrial interest. However, the selectivity and efficiency are not constant – they change significantly among different copper catalysts or among different regions of the same catalyst.
Using simulations based on the Density Functional Theory, Lucas and the other authors of the article tried to understand how the structural differences of the copper surface change the reactions involved in the transformation of CO2, affecting the performance of the catalysts. “Our study showed that bifurcations in the path of reaction can happen in a same type of catalyst (the copper one) changing structural properties (the level of coordination of atoms)”, says Lucas.
In addition to explaining the changes that were noted experimentally in the activity and selectivity of copper catalysts, the paper contributes to the development of the computational method that was used. Finally, the research results should help develop copper surfaces capable of generating specific products from CO2 in a controlled way.
The work, coordinated by professor Juarez L. F. Da Silva (IQSC-USP), researcher of the Computational Materials Science and Chemistry Division at CINE, was performed within the post-doctorate project of Lucas Verga with the collaboration of doctor Paulo C. D. Mendes and the PhD student Vivianne K. Ocampo – Restrepo.
Paper: The Role of Site Coordination on the CO2 Electro reduction Pathway on Stepped and Defected Copper Surfaces. L. Garcia Verga, P. C. D. Mendes, V. K. Ocampo-Restrepo and J. L. F. Da Silva, Catal. Sci. Technol., 2021, DOI: 10.1039/D0CY02337J.
Contact

Lucas Garcia Verga
USP, Brasil

